SoLiK Li-hochleitende Keramiken für all-solid-state Batterien
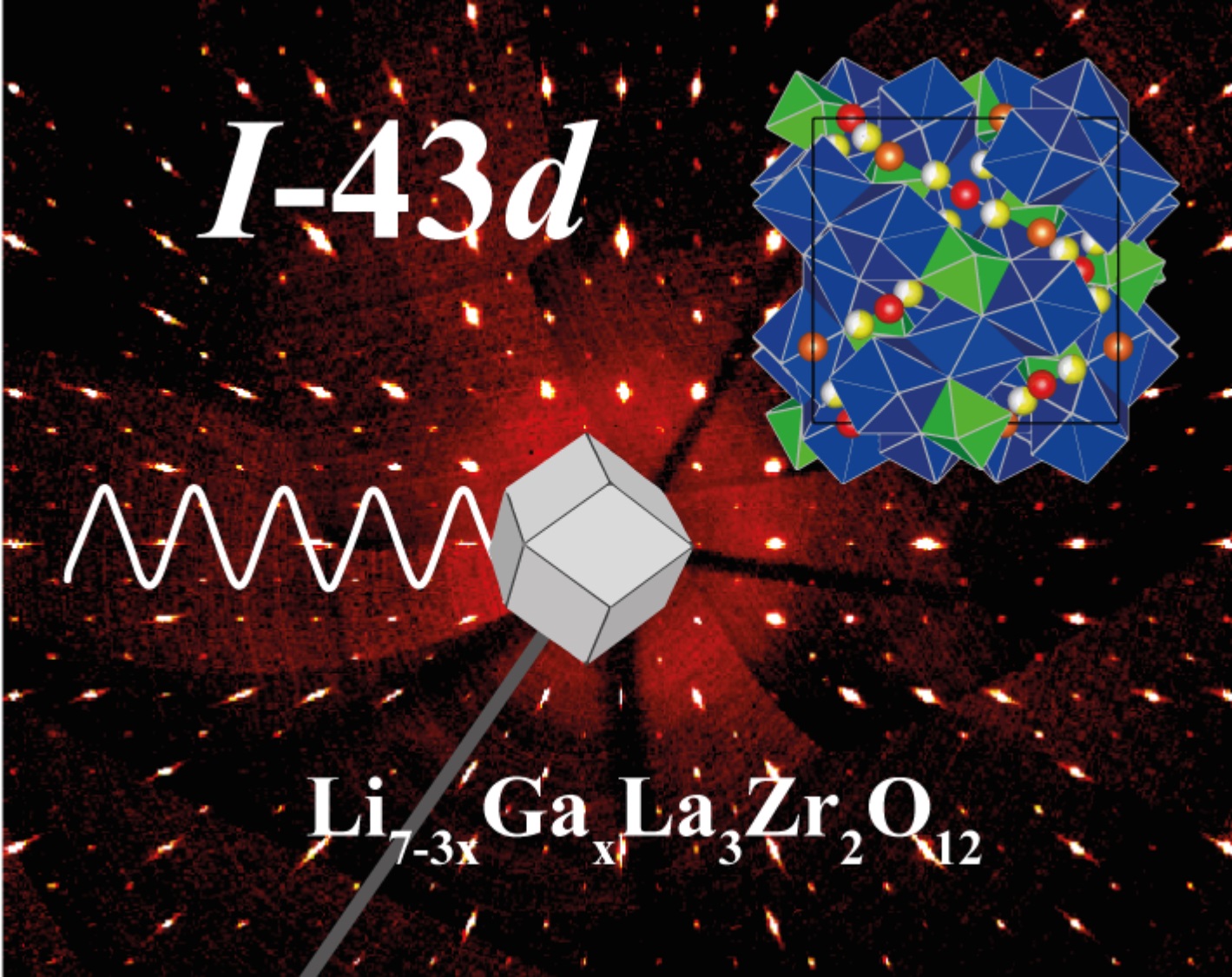
Lithium-oxide garnets, with a general composition of Li7La3Zr2O12(LLZO), present one of the highest Li-ion conductivities measured in solids. Thus, they are exceptionally suited for use as solid-state electrolytes in all-solid-state batteries, which are among the most important candidates for very safe and efficient, high-performing future energy storage systems.
Although much research on Li7La3Zr2O12 has been done in recent years, many important issues which impact the understanding of the connection between its crystal chemistry and Li-ion conductivity remain unsolved. Working toward a thorough understanding of the effective influence of doping on Li-ion transport in LLZO was a main goal of this project. The chosen approach included spatially resolved quantitative chemical analysis as well as structural and electrochemical characterization of the same materials. Also, some insight was gained on the influence of grain boundaries on total conductivity. Moreover, a new group of parameters was found to have a complex, sometimes even seemingly contradictory influence on conductivity: the concentration of oxygen defects in the LLZO crystal, which also varies with the doping elements introduced into the synthesis.
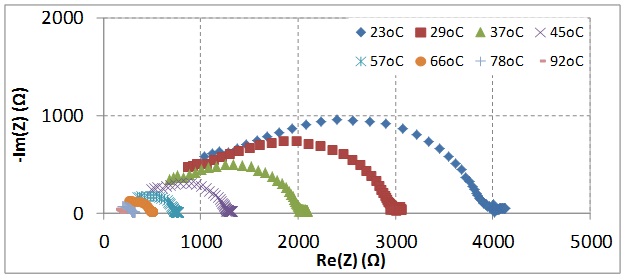
Electrical conductivity on grains and grain boundaries was measured locally by means of microelectrodes and correlated with precise, spatially resolved quantitative chemical analysis of the measured samples. The electrical conductivity in the LLZOs was investigated with Li-ion blocking as well as electrochemically active Li metal electrodes.
The project also included preparation of LLZO thin films and the conductivity in the films was studied by in-plane measurements.
The last phase of the project dealt with investigations into the preparation issues of sandwich-like designed all-solid-state batteries with LLZO-electrolyte and the cells were characterized electrochemically.
Ausgangssituation
All solid state batteries hold much promise in terms of safety and cost for many kinds of aplications, from wearable electronics to electric vehicles. So far, their large scale application has been hindered by issues of low conductivity, which leads to unfeasibly slow charge and discharge processes. One approach to understanding and hopefully improving this situation was followed in the SoLiK basic research project: introduce small amounts of other elements („doping“) and measure with unprecedented precision how the changed crystal chemistries affect conductivity.



Projektverlauf
The workflow in the SoLiK project was:
1. Synthesis of Al, Ga, Ta, and Nb doped LLZO garnet electrolytes via mixing oxides and sol-gel methods. The highest Li ionic conductivity was measured in the sample of LLZO-Ga0.2 pfu (per formular unit) to be ~1.2 mS/cm through the electrochemical impedance spectroscopy method.
2. Successful sintering of highly densified LLZO samples and LLZO samples with large grains -up to 1 mm- by optimizing sintering temperature, time, atmosphere, etc.
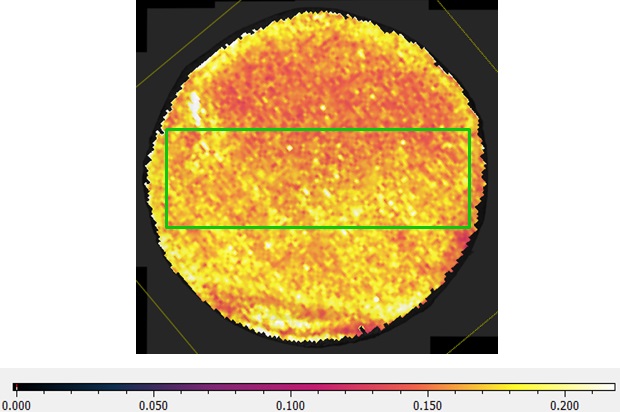
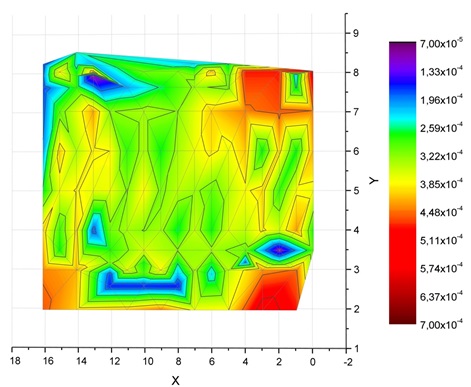
3. Microelectrode technology was successfullly applied to measure the local Li ionic conductivity in LLZO samples, and the distribuiton of local Li ionic conductivity in LLZO samples agreed well with the chemical distribution of Lithium element in the same sample determined by LA- ICP MS (Laser Ablation Induced Coupled Plasma Mass Spectroscope).
4. Densified and uniform LLZO-Ga0.2 thin films with a thickness of ~200nm were deposited on substrates of Si, SiO2, glasses, Al, and Au by magnetron sputtering method, and their crystalline structures, morphologies, and electrochemical properties were characterized by XRD, SEM, and EIS respectively.
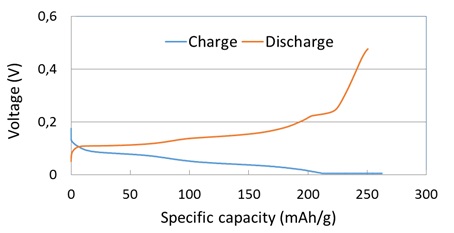
5. LLZO thick films from LLZO powders were prepared directly on commerical anode and cathode films to form the composite LLZO/anode and LLZO/cathode films, and all-solid-state lithium-ion batteries were prepared based on the composite LLZO/electrode films. Characterization of EIS and charging-discharging curves of the all-solid-state lithium-ion batteries prototypes gave a comprehensive picture of the materials‘ performance.
Meilensteine
- Synthesis of garnet structured LLZO ceramics doped with Al, Ga, Ta and Nb element with high density and varied grain sizes
- Phase and chemical analysis of bulk samples and single crystals
- Electrochemical analysis of the Li ionic conductivity of bulk samples with macro electrode and micro electrode
- Correlation of quantitative chemical analysis with the ionic conductivity of doped garnet LLZO samples
- Preparation of LLZO-Ga thin films via magnetron sputtering technology
- Phase analysis and ionic conductivity measurement of LLZO thin films
- Preparation of all-solid-state batteries based on thick film of LLZO-Ga and electrochemical analysis
"The stone age came to an end not for lack of stones, and the oil age will come to an end not for lack of oil."
– Sheikh Yamani, former Saudi Oil Minister –
Ergebnisse
Reasonable electrochemical performance was achieved for the thick film batteries when a very small amount of liquid electrolyte was added. We hope that in future projects the solid electrolyte can be made thin enough so that this addition is no longer necessary.
Since this was a basic science project, success is measured in scientific publications rather that in prototype performance.
An international workshop on solid lithium ion conductors was successfully held at TechGate, Vienna, on May 20, 2015, where several European scientists as well as the SoLiK consortium presented their researches on solid lithium electrolytes from theory to practical application, and shared their anticipation on the furture development of solid electrolytes for electrical energy storage.
As to the dissemination of the project results, seven articles have been or will be published in internationally renowned journals such as „Chemistry of Materials“, „Solid State Ionics“, the „Journal of Electroceramics“, etc. The project also yielded two successful master’s theses and two doctoral dissertations.
Several posters/presentations were or will be shown at international conferences on lithium ion batteries (ABAA9, Batteries Event 2017, and TRA2018, etc.).
Steckbrief
-
Projektnummer843882
-
Koordinator
-
ProjektleitungNingxin ZHANG, ningxin.zhang@ait.ac.at
-
Partner
-
SchlagwörterDünnschichtabscheidung, Elektrofahrzeug, Festkörperelektrolyt, Lithium-Ionen Batterie
-
FörderprogrammEnergieforschung (e!MISSION)
-
Dauer05.2014 - 04.2017
-
Budget701.409 €




